Study Materials
General Studies - Environment
Acid Rain & Ocean Acidification
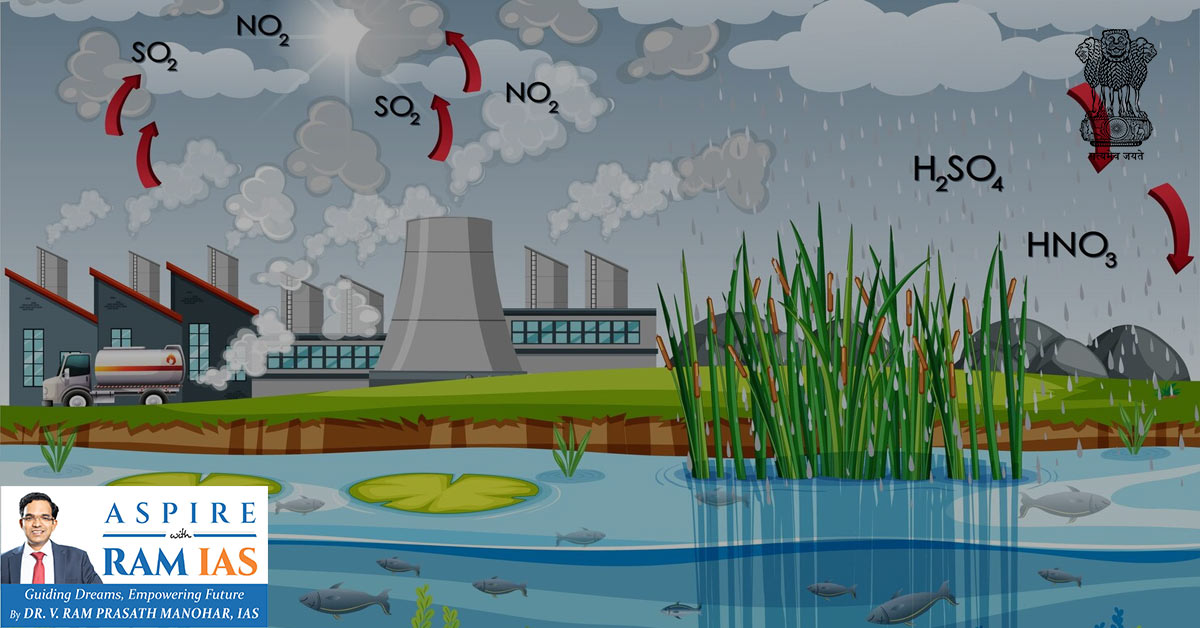
1. Acid Rain (Acid Deposition)
1.1 Meaning
- Acid rain refers to precipitation with a pH < 5.6
- Includes:
- Rain
- Snow
- Fog
- Mist
- Normal rainwater is slightly acidic due to carbonic acid (CO₂ + H₂O), but not acid rain
1.2 Causes
- Emission of acidic gases:
- Sulphur dioxide (SO₂ / SOx)
- Nitrogen oxides (NO, NO₂, N₂O collectively NOx)
- Released mainly from:
- Fossil fuel burning
- Thermal power plants
- Smelting industries
- Vehicular emissions
1.3 pH Scale (Prelims Favourite)
- Range: 0–14
- 7 → Neutral
- <7 → Acidic
- >7 → Basic
- Each unit decrease in pH → 10× increase in acidity
1.4 Gases Responsible for Acid Rain
Acidic Gases | Major Sources |
SOx | Coal burning, smelting, fertiliser industry, volcanoes |
NOx | Fossil fuels, power plants, lightning, biomass burning |
1.5 Types of Acid Deposition
A. Wet Deposition
- Acidic rain, snow, fog, mist
- Directly affects soil, crops, and lakes
B. Dry Deposition
- Acidic gases & particles settle on:
- Buildings
- Vegetation
- Soil
- Washed down later by rain → more acidic runoff
- Nearly 50% acidity comes via dry deposition
1.6 Chemistry of Acid Rain (Simplified)
- Emission of SOx & NOx
II. Sunlight forms photo-oxidants (e.g., ozone)
III. Oxidation reactions occur
IV. Formation of:
- Sulphuric acid (H₂SO₄)
- Nitric acid (HNO₃)
Acidic ions fall as wet/dry deposition
2. Harmful Effects of Acid Rain
2.1 On Human Health
- Eye, skin, and respiratory irritation
- Aggravates:
- Bronchitis
- Emphysema
- Lung cancer (indirectly)
2.2 On Soil
- Leaching of nutrients:
- Calcium
- Magnesium
- Potassium
- Release of toxic metals:
- Aluminium
- Lead
- Indian soils → mostly alkaline → better buffering capacity
2.3 On Aquatic Ecosystems
- pH change kills:
- Fish eggs
- Amphibian larvae
- Heavy metals are released into water
- Microbial productivity declines
2.4 On Terrestrial Plants
- Leaf cuticle damage
- Reduced photosynthesis
- Metal toxicity through roots
2.5 On Microorganisms
- Shift from:
- Bacteria-dominated → Fungi-dominated soil
- Slower decomposition
- Reduced soil fertility
2.6 On Buildings & Monuments
- Marble & limestone react with acids
- Causes:
- Flaking
- Black crust formation
- Example:
- Taj Trapezium Zone – “Marble Cancer”
2.7 Acid Rain Regions
- Global:
- Scandinavia
- NE USA
- Canada
- NW Europe
- India:
- First reported: Mumbai (1974)
- NE India, coastal Karnataka, Kerala, Odisha, WB, Bihar
2.8 Control Measures
- Low-sulphur fuel
- Washed coal
- Use of natural gas
- Buffering (Liming):
- CaO / CaCO₃ added to acidified lakes
3. Ocean Acidification
3.1 Meaning
- Ongoing decrease in ocean pH
- Due to the absorption of atmospheric CO₂
- Called:
- “Evil twin of global warming”
- “Other CO₂ problem”
3.2 Chemical Process
- CO₂ dissolves in seawater
II. Forms carbonic acid (H₂CO₃)
III. Produces:
- Bicarbonate (HCO₃⁻)
- Hydrogen ions (H⁺)
pH decreases → acidity increases
3.3 Major Contributors
- Fossil fuel emissions
- Deforestation
- Eutrophication:
- Plankton blooms → decomposition → CO₂ increase
4. Effects of Ocean Acidification
4.1 On Marine Chemistry
- Decrease in carbonate ions (CO₃²⁻)
- Difficulty in forming:
- Calcium carbonate (CaCO₃)
4.2 On Marine Life
- Affects calcifying organisms:
- Corals
- Molluscs
- Calcareous plankton
- Leads to:
- Coral bleaching
- Weak shells
- Reduced growth
4.3 On Fisheries
- Damage to the base of the food web
- Threatens:
- Arctic fisheries
- Commercial fisheries
4.4 On Climate Regulation
- Oceans absorb ~1/3rd anthropogenic CO₂
- Acidification reduces buffering capacity
5. Artificial Cloud Seeding
- Technique to induce rainfall
- Uses:
- Silver iodide
- Dry ice
- Encourages ice crystal formation in clouds
